Products
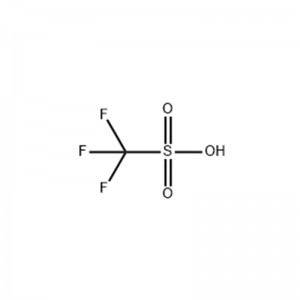
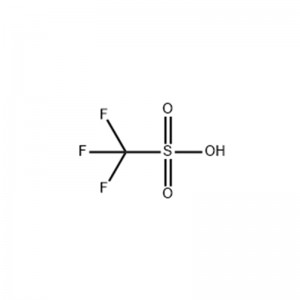
Trifluoromethanesulfonic Acid
Structural Formula
Physical Properties
Appearance: Yellowish brown liquid
Density: 1.696 g/mL at 25 °C (lit.)
Melting point: -40 °C
Boiling point: 162 °C (lit.)
Refractivity: n20/D 1.327(lit.)
Flash point: None
Acidity coefficient (pKa): -14 (at25 °C)
Specific gravity: 1.696
PH value: <1(H2O)
Safety Data
Belongs to the dangerous goods
Hazardous category: 8
Hazardous material transport number: UN 3265 8/PG 2
Packing group: II
Customs code:2904990090
Export Tax Refund Rate(%):9%
Application
It is the strongest known organic acid and a versatile synthetic tool. With strong corrosivity and hygroscopicity, it is widely used in pharmaceutical and chemical industries, such as nucleosides, antibiotics, steroids, protein, saccharides, vitamin synthesis, silicone rubber modification, etc. With small dosage, strong acidity and stable properties, it can replace traditional inorganic acids such as sulfuric acid and hydrochloric acid in many occasions and play a role in optimizing and improving the process. It can also be used as a catalyst for isomerization and alkylation to prepare 2,3-dihydro-2-indenone and 1-tetralone, and to remove glycoside from glycoprotein.
Safety Precautions
Trifluoromethanesulfonic acid is one of the most powerful organic acids. Contact with the eyes will cause severe eye burns and possible blindness. Contact with the skin will cause severe chemical burns, as well as delayed severe tissue damage. Inhalation of vapors can cause severe convulsive reactions, inflammation, and edema. Ingestion can cause severe gastrointestinal burns. Therefore, even small amounts require proper protective equipment (such as goggles, acid and alkali resistant gloves, and a gas mask), and good ventilation.
The addition of trifluoromethanesulfonic acid to polar solvents results in exotherm due to dissolution. This intense exotherm is similar to the effect of dissolving sulfuric acid in water. However, dissolving it in a polar solvent is inherently more dangerous than dissolving sulfuric acid in water. The strong exotherm may cause the solvent to evaporate or even explode. Therefore, dissolving large amounts of trifluoromethanesulfonic acid in organic solvents should be avoided. When it is necessary to do so, be sure to control the drop acceleration and ensure adequate stirring, good ventilation, and possibly cooling exchange devices to remove as much of the heat generated as possible.